Immunomodulatory Therapy in Pediatric Uveitis,
A comprehensive review up to 2022
Arash Maleki, MD; C. Stephen Foster, MD, FACS, FACR
April 2026
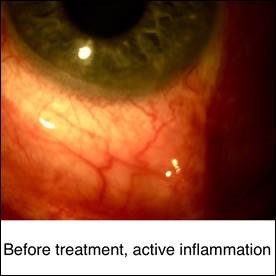
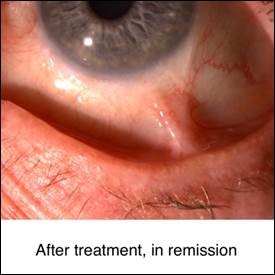
The primary goal of treatment is to suppress active inflammation and decrease the risk of complications. If the cause of uveitis is an infection, appropriate therapy for that infection should be employed with or without anti-inflammatory therapy. Currently, despite recent and ongoing clinical trials, the non-infectious uveitis treatment is based on expert opinion and algorithms proposed by multi-disciplinary panels. A multi-disciplinary approach between pediatric rheumatologists and uveitis specialists is the key for successful treatment. Although corticosteroids are still the first line of treatment of acute uveitis, the major long-term goal of therapy is corticosteroid-free remission with corticosteroid-sparing immunomodulatory therapy. A step ladder approach is commonly employed in the treatment of pediatric patients with uveitis. This approach starts with the use of less potent medications and medications with better safety profiles and shifts to more potent medications with more potential side effects in patients with insufficient response. During corticosteroid free immunomodulatory therapy, high risk blood monitoring tests should be performed at regular intervals. These blood tests include complete blood count (CBC), liver function tests (LFTs), BUN and Cr. Patients on immunomodulatory therapy with conventional medications and biologic response modifying agents should avoid vaccination with live viruses. Almost all conventional immunomodulatory therapies are teratogenic and should be used with caution in child-bearing age.
Corticosteroids
The first line of treatment in pediatric anterior uveitis is often topical corticosteroids. Difluprednate 0.05% is the most potent topical corticosteroid; however, prednisolone acetate 1% is the most commonly used topical corticosteroid in uveitis. While topical corticosteroids are preferred over systemic corticosteroid therapy in most cases, systemic (oral and/or intravenous) and local (periocular and/or intraocular) injections can be considered for resistant, intermediate, posterior, and panuveitis. Slow-release intraocular implants such as dexamethasone intravitreal implant which can last up to 6 months and fluocinolone acetonide 0.19 mg which last for 3 years, are available for more severe cases. Cataract and glaucoma are the most common potential side effects of steroids, especially with intravitreal corticosteroids implants. Oral corticosteroids are used in inflammation recalcitrant to local therapy and in bilateral uveitis. Oral prednisone, at a dose of 1-2 mg/kg of body weight, is the most commonly prescribed oral corticosteroid. However, long-term systemic corticosteroid use should be avoided, especially in the pediatric age group, due to serious side effects including growth retardation, weight gain, hyperglycemia, infection, and osteoporosis. For other types of chronic uveitis in pediatric patients, the recommendations from the American College of Rheumatology/Arthritis Foundation for glucocorticoid use in JIA are employed. These include, 1) superiority of prednisolone acetate over difluprednate topical drops, 2) superiority of adding or increasing topical corticosteroids over systemic corticosteroids for controlling active inflammation, 3) superiority of topical corticosteroid over changing or escalating systemic therapy in patients with flare up on systemic therapy, 4) superiority of adding systemic therapy for patients who need 1-2 drops for controlling the inflammation, 5) in children and adolescents with JIA and CAU still requiring 1–2 drops/day of prednisolone acetate 1% (or equivalent) for at least 3 months and on systemic therapy for uveitis control, changing or escalating systemic therapy is conditionally recommended over maintaining current systemic therapy.14
Non-steroidal Anti-inflammatory Drugs (NSAIDs)
Naproxen and tolmentin have the longest history of successful treatment in pediatric uveitis. It has been shown that celecoxib and diflunisal may be useful in controlling anterior uveitis associated with seronegative spondyloarthropathies. These are not typically the drug of choice for different types of vasculitis.
Conventional Immunomodulatory Therapy
Since long-term use of corticosteroid can incur serious side effects, corticosteroid-sparing immunomodulatory therapy should be the next step in the stepladder approach for the treatment of pediatric uveitis. Conventional immunomodulatory therapies include methotrexate and less commonly used mycophenolate mofetil, azathioprine and cyclosporine. Methotrexate, mycophenolate mofetil, and azathioprine are all anti-metabolites while cyclosporine is considered a T-cell inhibitor. Anti-metabolites and T-cell inhibitors have additive effects on each other; however, anti-metabolites should not be used together preferably.
Methotrexate
Methotrexate is the most frequently used steroid-sparing agent in pediatric uveitis. Methotrexate is considered the first line of therapy in most cases of chronic non-infectious uveitis. The recommended starting dose is 10-15 mg/m2 once a week orally or subcutaneously. Maximum dose is 30 mg/m2; however, doses higher than 15 mg/m2 should be used subcutaneously due to the limited oral bioavailability of methotrexate. A meta-analysis demonstrated that methotrexate was effective in controlling pediatric non-infectious uveitis and the proportion of responding subjects was 0.73 in nine eligible studies. Common side effects of methotrexate include liver, renal and gastrointestinal toxicity. This medication should be used with folic acid supplements to help with normal cell proliferation and prevent side effects.
Mycophenolate Mofetil (MMF)
The optimal dose of MMF for uveitis is unknown; however, the therapy usually starts at a dose of 600 mg/m2 twice a day and the max dose is 3 g/day in two divided doses. There are two reports of MMF therapy in pediatric patients. Although MMF is helpful in the treatment of non-infectious uveitis, it is less effective for arthritis in comparison with other agents. Leukopenia, gastrointestinal discomfort, and hair loss are major side effects.
Azathioprine
Azathioprine can be moderately effective in the pediatric population; however, it is less commonly used due to the increased risk of gastrointestinal side effects in comparison to other anti-metabolites. There are few case series which report the experience with azathioprine in JIA associated uveitis. An observational multi-center study showed that azathioprine may be helpful as a systemic monotherapy or in combination with other immunosuppressive agents in JIA associated uveitis. The safe and well-tolerated dose in children is 3 mg/kg in a single dose or divided dose.
Cyclosporine
It is important to note that cyclosporine is minimally effective as monotherapy; however, the efficacy of cyclosporine increases when it is combined with other immunomodulatory agents. Pediatric dosage of cyclosporine is 2.5 to 5.0 mg/kg/day in divided doses to prevent drug toxicity. Nephrotoxicity, hypertension, hepatotoxicity, hypercholesterolemia, and hirsutism are potential side effects. The dosage of cyclosporine is adjusted based on the medication level. Cyclosporine is infrequently employed in the treatment of uveitis in children.
Biologic Response modifiers
Biologic response modifiers are the next step of the stepladder approach after conventional immunomodulatory agents. These agents block target molecules in the immune system that play a role in the ocular inflammatory process. Therapeutic options include anti-tumor necrosis factor alpha (TNF-α), anti-interleukin 1 (IL-1), anti-B-cell, and anti-T-cell inhibitors. Viral hepatitis and tuberculosis should be ruled out before starting these agents. Out of all the biologic response modifier agents, anti-TNF-α agents have been studied most frequently.
Tumor Necrosis Factor Alpha Inhibitors (TNF-α inhibitors)
Infliximab
Infliximab is a chimeric (human-murine) monoclonal antibody that binds and inhibits circulating and membrane bound TNF-α. The efficacy and safety of infliximab have been studied in retrospective and prospective studies. Infliximab is administrated parenterally with a loading dose of 3-5 mg/kg at 0-2-6 weeks, followed by a maintenance dose of 3-10 mg/kg every 4 weeks. Low dose methotrexate is recommended to prevent antibody formation against the murine part of infliximab molecule. In refractory non-infectious uveitis, a higher dose of 20 mg/kg has been used successfully. Major side effects include susceptibility to infections, reactivation of tuberculosis and histoplasmosis, malignancy, and the development of lupus-like syndrome. Mild to moderate infusion reactions can be seen in 17% of patients.
Adalimumab
Adalimumab, a fully humanized monoclonal antibody, is the only biologic response modifier agent that has been approved by the U.S. Food and Drug Administration (FDA) for the treatment of non-infectious uveitis. Several studies have supported the efficacy of adalimumab in the treatment of pediatric uveitis. Antibody development against adalimumab has been reported in previous studies (13%). This is a favorite medication for ocular immunologists in the treatment of pediatric uveitis. Adalimumab is administered subcutaneously at a dose of 24 mg/m2 up to maximum dose of 40 mg every 2 weeks; however, this interval can be shortened to weekly when the response is inadequate. It was demonstrated in a multicenter, double blind, randomized, placebo-controlled trial (SYCAMORE Trial) that adalimumab therapy-controlled inflammation and was associated with a lower rate of treatment failure than placebo among children and adolescents with active JIA-associated uveitis on a stable dose of methotrexate.
Adalimumab was also shown to be effective in cases of early onset, chronic anterior uveitis with inadequate response to topical therapy and methotrexate.
A meta-analysis performed by Maccora et al. showed the efficacy of adalimumab and infliximab in the treatment of chronic childhood uveitis. However, the results from adalimumab use were superior to those associated with infliximab use in this clinical setting.
The side effect profile of adalimumab is similar to that of infliximab. The high efficacy in pediatric uveitis and ease of administration subcutaneously at home make it a good choice for both the patient and the family.
Abatacept, a T-cell inhibitor biologic agent
Abatacept is a soluble fusion protein which binds to CD80/CD86 on antigen presenting cells and prevents T-cell activation. A recent study described abatacept efficacy in 3 children with idiopathic uveitis. In a study by Tappenier and colleagues, 48% of patients on abatacept failed treatment and it was concluded that abatacept was not a successful treatment in pediatric uveitis. Dosing is 10 mg/kg at 0 and 15 days followed by monthly infusion. Alternatively, abatacept can also be employed as a subcutaneous injection at a dose of 125 mg weekly. Side effects include infection susceptibility, gastrointestinal symptoms, and low risk of malignancy. The efficacy of abatacept in pediatric uveitis is inconclusive.
Tocilizumab, an interleukin-6 (IL-6) inhibitor
Tocilizumab is a humanized recombinant anti-IL-6 receptor antibody which can inhibit T-cell activation, immunoglobulin secretion, and angiogenesis. We recently studied tocilizumab on 8 patients (14 eyes) with refractory JIA associated uveitis. The average duration of follow-up period after starting treatment was 28.6 ± 24.6 months (9-70). Intravenous tocilizumab infusions induced and maintained remission in 5 patients (8 eyes). Vasculitis was resolved within 8 months in all but one patient. We found that intravenous tocilizumab infusion can be an effective and safe method of treatment to induce and maintain remission in resistant JIA associated uveitis. Ramanan et al. studied tocilizumab in patients with anti-TNF refractory juvenile idiopathic arthritis associated uveitis (APTITUDE trial). The aim of this phase two trial was to study the safety and efficacy of tocilizumab in children with juvenile idiopathic arthritis-associated uveitis refractory to both methotrexate and TNF inhibitors; however, they did not meet the primary endpoint of their phase 2 trial to support a phase 3 trial of tocilizumab in patients with JIA-associated uveitis. Currently, there are two ongoing trials to study the efficacy of tocilizumab in noninfectious intermediate, posterior and panuveitis: (STOP-Uveitis) and JIA-associated uveitis (JIA-U). (www.clinicaltrials.gov) Tocilizumab has been proposed to be a promising agent for the treatment of pediatric uveitis; however, more studies are required for the ideal dose and interval. The current dose of medication is 165 mg/week through subcutaneous injections or 8 mg/kg of weight through monthly infusions. Monthly infusions of tocilizumab have been found to be more effective than weekly injections.
Rituximab, a B-cell inhibitor biologic agent
Rituximab is a chimeric antibody that targets the B-cell marker CD20 and subsequently induces B-cell apoptosis. Other mechanisms of action include receptor signaling, calcium flux, complement activation or attraction of antibody-dependent effector cells. Miserocchi and colleagues studied a cohort of 8 patients (15 eyes) with severe refractory JIA uveitis treated with rituximab. The mean number of infusions was 8.75 during mean 47.5 months follow-up (almost one infusion every 5 months). Uveitis was inactive in all patients at the last follow-up visit, and improvement was noted 4 months after the first infusion.
Rituximab is traditionally infused at a dose of 17 mg/kg per infusion which would result in a total dose of 70 mg/kg (4 weekly infusions). The long-lasting effect (6-9 months) of rituximab make it an attractive biologic agent. Rituximab in pediatrics is traditionally given slowly, administered at an initial rate of 0.5 mg/kg/hr for the first hour, and is gradually increased by 0.5 mg/kg/hr every 30 minutes based on the patient’s response (maximum rate of 50 mg/hr).
Side effects include infusion reaction, late neutropenia, hypogammaglobulinemia, heart failure, and rarely lethal progressive multifocal neuroencephalopathy (PMN). IgG monitoring may be useful for patients receiving rituximab since persistent hypogammaglobulinemia that required IgG replacement was noted in 4.2% of patients.
Emerging Biologics
Janus Kinase (JAK) inhibitors have been recently employed in the treatment of patients with uveitis and scleritis in adults. There is an ongoing open-label, active-controlled, safety, and efficacy study of oral baricitinib in patients from 2-18 years old with active JIA-associated uveitis or chronic anterior ANA-positive uveitis.
The recommendation of the American College of Rheumatology/Arthritis Foundation for immunomodulatory therapy in JIA which are mostly applied to other types of chronic uveitis in pediatric group include, 1) subcutaneous methotrexate is superior over oral methotrexate for starting treatment, 2) for a sight-threatening condition a combination of methotrexate and a biologic is recommended for the starting treatment, 3) superiority of other TNF-α inhibitors over etanercept in chronic anterior uveitis (etanercept in ineffective in ocular inflammatory diseases), 4) TNF-α inhibitors dose should be boosted and/or interval should be shortened in inadequate response to standard dose of TNF-α inhibitors, 5) second TNF-α inhibitor should be employed after failing the first one, 6) abatacept and tocilizumab can be employed in patients who fail methotrexate and two TNF-α inhibitors, and 7) all effective medications should be continued for 2 years before tapering therapy.
References
Maleki A, Anesi SD, Look-Why S, Manhapra A, Foster CS. Pediatric uveitis: A comprehensive review. Surv Ophthalmol. 2022;67(2):510-529.